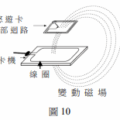
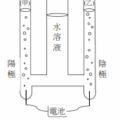
<多選題>已知下列三個反應式與其反應熱如下:
\[2N_2(g) + 6H_2(g) \rightarrow 4NH_3(g) \quad \Delta H_1\]\[2N_2(g) + 6H_2(g) + 7O_2(g) \rightarrow 4NO_2(g) + 6H_2O(g) \quad \Delta H_2\]\[4NH_3(g) + 7O_2(g) \rightarrow 4NO_2(g) + 6H_2O(g) \quad \Delta H_3\]則下列關係式中,哪些正確?
(A) \(\Delta H_2 = \Delta H_1 + \Delta H_3\)
(B) \(\Delta H_2 = \Delta H_1 – \Delta H_3\)
(C) \(\Delta H_1 = [NH_3(g)]\)的莫耳生成熱\(^1 \times 4\)
(D) \(\Delta H_2 = [NO_2(g)]\)的莫耳生成熱\(^1 \times 4\)
(E) \(\Delta H_3 = [NO_2(g)]\)的莫耳生成熱\(^1 \times 4 + [H_2O(g)]\)的莫耳生成熱\(^1 \times 6\)
– \([NH_3(g)]\)的莫耳生成熱\(^1 \times 4\)
答案